|
The atomic orbitals that combine are of similar energy levels a 1s orbital does not combine with one of the 2 s orbitals.\): Two atomic orbitals (left) will overlap to form bonding (b) and antibonding (ab) molecular orbitals. Similarly 2 s atomic orbitals combine, giving a bonding orbital and an antibonding orbital, which are filled with the remaining valence electrons starting from the bottom up. For the ion Li2:a) Draw the molecular orbital diagram.b) Calculate the bond order.c) Would this ion existd) Write the electron configuration of the ion.Inte. Notice that we have combined the 1 s atomic orbitals, as before in the H 2 example, to generate bonding and antibonding molecular orbitals that are completely filled by both atoms’ 1 s electrons. 0:15 Molecular Orbital Diagram of Hydrogen Molecule1:39 Molecular Orbital Diagram of Helium Molecule2:54 Molecular Orbital Diagram of Lithium Molecule4:00 Mo. Molecular orbital electron configuration energy diagram for dilithium. Learn how to draw a molecular orbital diagram for Li2, a diatomic molecule with six electrons, using the molecular orbital theory (MO theory). Each horizontal line represents one orbital that can hold two electrons. Lets follow these guidelines and generate a molecular orbital electron configuration diagram for Li2 (Figure 9.21 Molecular orbital electron configuration. - YouTube How to Make the Molecular Orbital Diagram for Li2: Bond Order WebExpress the bond order for Li2 followed by. This is a bond between two lithium atoms, which have an. Molecular Orbital Theory (MOT) was first proposed by Friedrich Hund and Robert Mulliken in 1933. Bond-order usually predicted from the Molecular Orbital Theory 1,2. 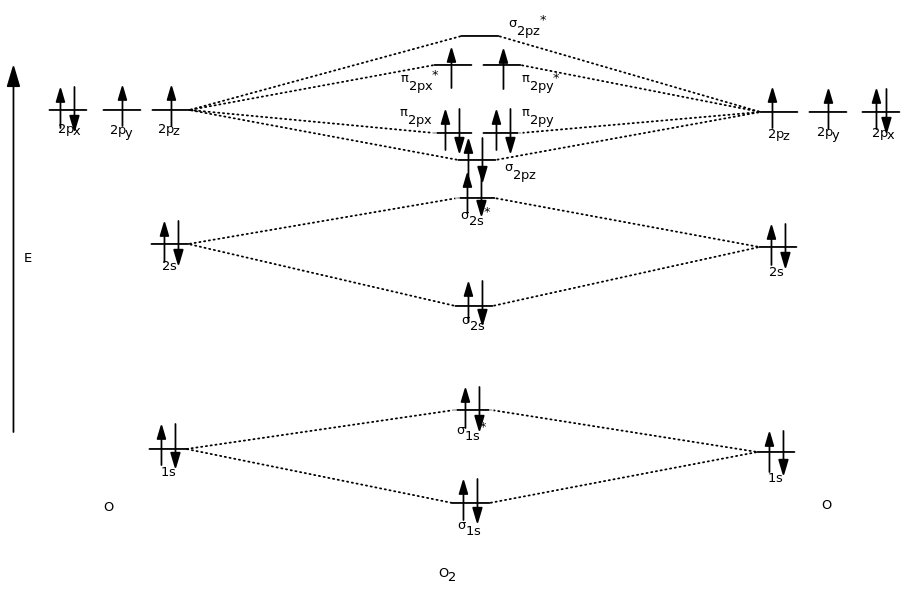
For a diatomic molecule, the atomic orbitals of one atom are shown on the left, and those of the other atom are shown on the right. Bond order of lithium molecule Consider the MO diagram for Li2. Predicting the Bond-Order of Diatomic Species. Let’s follow these guidelines and generate a molecular orbital electron configuration diagram for Li 2 (Figure 9.21 “Molecular orbital electron configuration energy diagram for dilithium”):įigure 9.21. The relative energy levels of atomic and molecular orbitals are typically shown in a molecular orbital diagram (Figure 8.34). Increased overlap lowers the energy of the bonding molecular orbital further, and raises the energy of the antibonding molecular orbital. – The effectiveness of atomic orbital combination depends on the amount of orbital overlap. Transcribed Image Text: Complete the atomic orbital (AO) and. Drag the appropriate labels to their respective targets. Assume the left AO comes from Li and the right AO comes from Li. Transcribed Image Text: Complete the AO and MO energy diagram for Liz. Using molecular orbital theory, determine the bond order for Li2. Use the MO diagrams to calculate the bond order for Li2+ and Li2. 
– Combined atomic orbitals should be of similar energy levels. molecular orbital energy-level diagram for MOs constructed from 1s atomic orbitals. 
– The number of MOs generated is equal to the number of atomic orbitals combined. Generating molecular orbitals of molecules more complex than hydrogen using the LCAO method requires following a few additional guidelines:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
